It is a fair, even-handed, noble adjustment of things, that while there is infection in disease and sorrow, there is nothing in the world so irresistibly contagious as laughter and good-humour.
Charles Dickens
The immune system basically describes the collection of biological processes within an organism that serve to protect it against disease by identifying and killing pathogens.
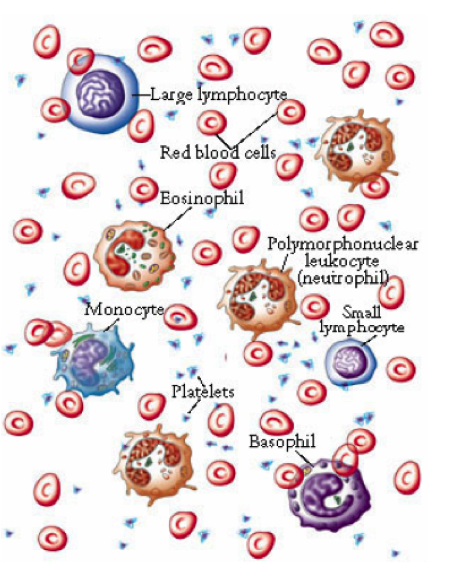
We have a special type of immune response known as the adaptive immune response that allows the immune system to recognize and remember specific pathogens (to generate immunity), and to mount stronger attacks each time the pathogen is encountered. Phagocyte cells in the blood are able to engulf antigens (foreign cells/proteins) and display them on their cell surface (defects in this can lead to chronic granulomatous disease). Other cells found in the blood, known as T-lymphocytes (defects in these ca lead to severe combined immunodeficiency syndrome) then recognise these foreign proteins and release chemicals to further break down this foreign material and also release additional molecules that attract and stimulate the division of further immune cells. These antigens also bind to the cell surface of another type of immune cell, B-lymphocytes (B-cells), causing these cells to divide and produce antibodies (immunoglobulins; Ig). Antibodies then function to destroy antigens by causing them to precipitate or clump, and can in addition initiate further pathways that cause the destruction of the antigen and stop the antigen from entering host cells. Finally, some of the activated B cells and T cells then go on to become memory cells, dividing and reproducing the same antigen through the individual’s lifetime. These B and T lymphocytes form part of the immune response, as these memory cells are able to respond extremely quickly to an antigen if it is recognised from the previous infection.

Perhaps the earliest documented incidence of acquired immunity was during the plague of Athens in 430 BC. Here it was noted at the time that people who had recovered from a previous bout of the disease could nurse the sick without contracting the illness a second time. This forms the basis of vaccination whereby a substance is introduced into the body in order to produce immunity to a disease.
Although similar procedures may have been used in China and India in ancient times, the first recorded vaccination (Lat. vacca; cow), was by Edward Jenner in 1796 when he used cowpox to give immunity to smallpox after observing that dairymaids who had previously been sick with cowpox (a contagious disease that causes blisters on the cow’s udder and on the milkmaid’s hand) did not catch smallpox. Pasteur then further developed the use of artificial vaccines that could be used for a variety of diseases such as rabies by growing the virus in dogs and rabbits, then weakening it by drying the affected tissues, and finally reintroducing it into healthy animals. This serves to activate an immune response and form an immune memory so that when the animal comes in contact with the infectious form of the virus its immune system can quickly respond.


Before the advent of antibiotic drugs most babies born with inherited immune defects would have died early due to their susceptibility to infections and such cases would not have been easy to identify among the many normal infants who would have also died of infections. It was not until 1952 that the first immunodeficiency disease was described, by Dr. Colonel Ogden C. Bruton, when he reported the failure of a male child to produce antibodies due to a defective gene hindering B-cell growth. This is now known as Bruton’s X-linked agammaglobulinemia, as the gene involved locates to the X chromosome. Dr Bruton also became the first physician to provide specific immunotherapy for this disorder by administering injections of antibodies to the patient who responded well to the treatment. Since then, many more diseases of antibody production have been described, where affected infants usually develop recurrent infections with pyogenic, or pus-forming, bacteria such as pneumonia-causing Streptococcus pneumoniae.
The most common inherited immunodeficiency is selective IgA antibody deficiency, which may be present in 1 in 400 people. While it seems to result in no specific symptoms it is found more often in people with chronic lung disease. Apart from IgA deficiency, the overall incidence of primary immunodeficiency is around 1 in 10,000.
While deficiencies in the immune system can lead to infections, an overactive immune system can lead to another class of diseases known as autoimmune diseases. This is characterised by a decreased ability of the immune system to distinguish foreign material from host cells resulting in the immune system attacking the individual’s own tissues.
Severe Combined ImmunoDeficiency
Patients with defects in T-cell development are highly susceptible to a broad range of infectious agents, reflecting the crucial role T-cells have in many parts of the immune system. Such diseases are known as Severe Combined ImmunoDeficiency (SCID) as patients, unable to generate any immunological memory or cell-mediated immune responses, suffer from infections from many opportunistic pathogens. This results in early death if untreated with either bone marrow transplantations or enzyme replacement therapy. The most common type of SCID is called XSCID due to the mutated gene being found on the X chromosome. This gene normally codes for part of a receptor on the surface of lymphocytes allowing them to mature, proliferate and mobilize to fight infections.
Awareness of these diseases was raised in the 1970s by the plight of David Vetter, who lived nearly all of his 12 years of life inside a sealed plastic bubble designed to protect him from infections. David Vetter’s parents first had a daughter, Katherine, who was normal and then a son who died six months after birth from SCID. They realised that any subsequent child would have a 50 percent chance of inheriting the same condition but believing that a bone marrow transplant from their healthy daughter would give any infected child a normal healthy life, they decided to go through with another pregnancy. Twenty seconds after being removed from his mother’s womb, in September 1971, David Vetter was sealed in a germ-free environment that would be his home for nearly all of the rest of his life. Water, air, food, diapers and clothes were all disinfected with special cleaning agents before entering his cocoon and he was handled only through special plastic gloves attached to the walls. Eventually, a bone marrow transplant was performed using marrow donated by his sister Katherine. However, a few months after the operation, David started having diarrhoea, fever and severe vomiting, requiring him to be taken out of the bubble for the first time in 1984, for treatment. Sadly though, he died 15 days later of a lymphoma, caused by an unscreened virus, in his newly transplanted bone marrow.
Chronic Granulomatous
Generally X-linked
1:200,000
Phagocytic (Gr. phage; eat) cells destroy foreign matter, such as microorganisms by ingesting them into their cells where various enzymes reactive oxygen-containing molecules are present to break down the pathogen. There are a number of gene mutations that can lead to defects in the ability of phagocytes cells to kill pathogens. This occurs in chronic granulomatous disease due to mutations in a number of different genes resulting in recurrent chronic bacterial infections.
Autoimmune Diseases

Autoimmune diseases are characterised by a decreased ability of the immune system to distinguish foreign material from host cells resulting in the immune system attacking the individual’s own tissues. There are more than forty different autoimmune diseases affecting 5% to 7% of the population. Women account for a greater proportion of the sufferers – nearly 80% in the USA – which may be due to the role of sex hormones.
Some people are born with an inherited genetic susceptibility to autoimmune diseases. Such genetic risk factors often involve the inheritance of specific variants of genes called Human Lymphocyte Antigens (HLA). The function of these HLA proteins, displayed by all cells of the body, is to present fragments of antigens, either from the host cell or from invading pathogens on the outside of the cell for passing T-cells to recognise.
Depending upon which tissue in the body the immune system attacks, a number of different diseases can develop. For example, the production of antibodies against tissues of the thyroid gland can lead to over manufacture of thyroid hormone resulting in hyperthyroidism (an overactive thyroid) seen in Grave’s disease. President George H. W. Bush was diagnosed in 1991 shortly after his wife Barbara and their dog Millie. Because of the remarkable coincidence of all three cases of auto-immune disease in one household, it was suspected that this may have been the result of a plot to poison the President and the Secret Service went as far as to test the water in all of the presidential residences, including the White House, for any toxins. Many pictures of Barbara Bush during this period show that she had developed many of the classical characteristics of the disease, such as a bulging of the eyes and a throat goitre.
It is thought that the wife of Vladimir Lenin, Nadezhda Krupskaya, also suffered from Graves’s disease with her bulging eyes giving her the Bolshevik codename of Fish. Furthermore, since Graves Disease can also disrupt the menstrual cycle, it is believed that this is why the couple never had children. The British comedian Marty Feldman was also famous for his bulging eyes.



The body may also start to produce an antibody against specific cell receptors, such as neurotransmitters in the disease Myasthenia Gravis. This has the effect of blocking nerve signals to muscle cells resulting in muscle weakness. Suzanne Rogers, an American actress best known for her role in the daytime television series “Days of Our Lives”, was diagnosed with Myasthenia Gravis in 1984, and insisted that her character, Maggie, be diagnosed with the same disease on the show in order to raise awareness of the condition.
When the body’s immune system attacks the myelin sheath surrounding nerve cell axons, multiple sclerosis (MS) occurs. Translating as “multiple hardenings”, the most popular hypothesis as to the cause of MS is that it results from an infection which primes a susceptible immune system to destroy myelin later in life. The body then tries to repair the damage with hard plaques of scar tissue which further disrupts the flow of electrical impulses. Although not considered a hereditary disease, genetics does play some role in determining susceptibility to MS, particularly the inheritance of certain HLA variants. The contribution of genes to MS is evident from studies in identical twins where the likelihood that the second twin may develop MS if the first twin does is about 30%. Moreover, there are some populations, such as the Inuit and the Japanese who have very low incidences. The earliest person to have a direct diagnoses of MS was Sir Augustus Frederick d’Este, grandson of George III, who left a highly detailed diary describing his 22 years living with the disease. His symptoms began at the age of 28 when he began to develop weakness of the legs, clumsiness of the hands, numbness, dizziness and bladder disturbances that worsened until 1844, when he was confined to a wheelchair. The American comedian, Richard Pryor, in his later years became a wheelchair user due to MS, which he said stood for More Shit.


While some autoimmune diseases are due to an immune response to a specific tissue, others are characterised by general tissue damage throughout the body, i.e. systemic. This is seen in systemic lupus erythematosis where antibodies are produced to cells in many tissues of the body causing the immune system to attack and damage various parts of the body including the joints, skin, kidneys, heart lungs, blood vessels and brain. Research indicates that systemic lupus erythematosis has some genetic link and can run in families, with several genes identified, however, symptoms and severity vary enormously between individuals. The term lupus is attributed to the 12th century physician Rogerius, who used it to describe the classic reddish, butterfly-shaped rash across the nose and cheeks – the name either deriving from the Latin for wolf as it can resemble the pattern of fur on a wolf’s face, or from aFrench style of mask which women reportedly wore to conceal the rash on their faces. The English singer/songwriter, Seal, was diagnosed with the skin-affecting variant of this condition as a young man which has left him with the distinctive facial scars on his cheeks. Other singers, Lady Gaga, Elaine Paige, Kéllé Bryan and Toni Braxton are also reported to suffer.
