HISTORY OF GENETICS

‘My scientific studies have afforded me great gratification; and I am convinced that it will not be longbefore the whole world acknowledges the results of my work.’ – Gregor Mendel
Gregor Mendel
It was an Austrian Monk who first laid down the laws of genetic inheritance. In the 1860s he noticed that if pea plants producing purple flowers were pollinated by white flowered pea plants, the resulting seeds all produced plants with purple coloured flowers. Next, Mendel crossed the offspring of this purple-white flower cross (all of which had purple flowers) with each other. This resulted in a population (known as an F2 generation) of plants in which three-quarters produced purple flowers and one-quarter produced white flowers, i.e. a 3:1 ratio.
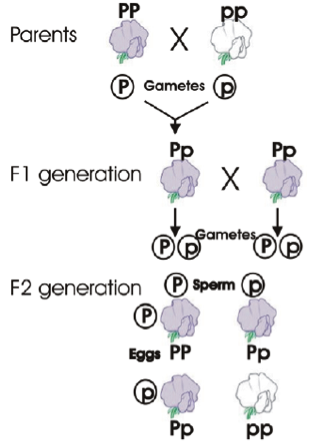
From this data Mendel developed his laws of inheritance, particularly the Law of dominance; when an organism has two different variants (we now use the term alleles) for a trait, the allele that is expressed, overshadowing the expression of the other allele, is said to bedominant (i.e. the purple flower – denoted by P in the diagram). The gene whose expression is overshadowed is said to be recessive (i.e. the white flower – denoted by p in the diagram).
Mendel published his findings in 1866 in the little-known journal Proceedings of the Natural History Society of Brunn. Even though these were possibly the most exciting findings ever in the field of science, signalling the birth of genetics, they had little impact being cited only three times over the next 35 years.
It was not until 1900 that other scientists rediscovered his work. Hugo de Vries published his findings, neglecting to mention Mendel. This prompted Carl Correns (a student of the renowned botanist Nägeli, whom Mendel corresponded about his work with peas but who failed to understand its significance) to remind de Vries about Mendel. Coincidently, at the same time, Erich von Tschermak, the grandson of Mendel’s old botany, also produced supporting findings. However, Mendel had long since died in 1884 from chronic nephritis.

Friedrich Miescher
The first person to isolate what we now know as DNA, or nucleic acid, was the Swiss scientist, Friedrich Miescher. Born in Basel to Friedrich Miescher-His, professor of pathological anatomy at Basel University, he decided to pursue a career in basic research rather than as a physician. This was due to the fact that he was quite hard of hearing and worried that he would be unable to use a stethoscope properly.
In 1868, he went to the physiological chemistry laboratory of Felix Hoppe-Seyler at Schloss Hohentübingen, who had just discovered the binding of oxygen to hemoglobin in erythrocytes.

Hoppe-Seyler quickly put Miescher to work on other blood cells, the lymphocytes from which he hoped to extract the nuclei, the small body inside the cell which we now know contains DNA. Lymphocytes are components of pus, which he obtained from the dirty bandages at a nearby hospital. But these cells were notoriously difficult to obtain. So tried to overcome the problems of washing the cells off the bandages without damaging them and isolating the nuclei by testing a series of different salt solutions before eventually producing one with sodium sulfate. While trying to isolate nuclei free of cytoplasm, he subjected purified nuclei to alkaline extraction followed by acidification resulting in a precipitate being formed, that he called “nuclein” i.e. this came from the nucleus.
Meisner asked Professor Hoppe-Seyler if he could publish the findings in the journal of which Hoppe-Seyler was editor. He provisionally accepted it, though would spend the next 2 years constantly repeating Miescher’s laboratory experiments before finally allowing it to be published.
Miescher then returned to Basel in 1870 and 2 years later was appointed to the newly-created chair of professor of physiology in Basel at the exceptionally young age of 28. And of course there was gossip that his family ties helped get him the appointment. Certainly, he worked hard to overcome this and in 1885, Miescher contracted pleurisy, perhaps because of the long hours spent in his ice-cold laboratory, when he neglected his ill health to the extent of not taking any nourishment for long periods. In 1894, he was diagnosed as suffering from tuberculosis. He died of the disease in a sanatorium in Davos in 1885, at age 51. As quoted by Portugal and Cohen, his former teacher, C. Ludwig, wrote to him shortly before his death: ”I know what it is to give up well-loved, hopeful work. Sad as it is, there remains for you the satisfaction of having completed immortal studies in which the main point has been the knowledge of the nucleus; and so, as men work on the cell in the course of the following centuries, your name will be gratefully remembered as the pioneer of this field.” Prophetic words, indeed !

‘Omnis nucleus e nucleo.’ – Walther Flemming
Walther Flemming
Using a red aniline dye, a by-product of coal tar, the German scientist Walther Flemming, stains the cells of salamander embryos. There he noticed red threadlike material that differently distributed and doubled during cell division. He named these chromosomes (Gr. Chrom; coloured body) and termed the process of cell division as mitosis (Gr. Mitos; thread, referring to the spindly chromosomes).
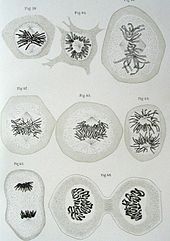
Remember that Mendel’s work had still not been read at this time and so Flemming’s work also receives little attention until ~20 years later where it is now recognised as one of the greatest scientific discoveries.

‘The world has been evolved, not created: it has arisen little by little from a small beginning, and has increased through the activity of the elemental forces embodied in itself, and so has rather grown than come into being at an almighty word.’ – August Weismann
August Weissmann
August Weismann is often credited as being among the all-time greatest thinkers and was one of the first to support Darwin’s theory of evolution against the idea of Christian creationism and was one of the staunchest supporters against the inheritance of acquired traits.
Exasperated by the continued belief that traits squired during life were inherited he devised an experiment to finally dispel the myth.

He set about chopping off over a thousand tails of generations of mice each time inspecting any litters for the birth of tailless mouse pup. He was able to report that not a single such animal inherited their parents amputated tail.
This provided further support for his “germ plasm” theory. Inheritance only takes place by means of the germ cells (i.e. egg and sperm) – somatic cells (any other biological cell of the body) do not function as agents of heredity. The effect is one-way: germ cells produce somatic cells and are not affected by anything the somatic cells learn or therefore any ability the body acquires during its life. He devised many of these ideas long before Mendel Mendel and was able to use this theory to directly support Darwin’s work.

‘Scientific method is not the same as the scientific spirit. The scientific spirit does not rest content with applying that which is already known, but is a restless spirit, ever pressing forward towards the regions of the unknown.’ – Archibald Garrod
Archibald Garrod
Archibald was the son of Sir Alfred Baring Garrod who was famous for discovering that abnormal uric acid metabolism associates with gout.
Archibald studied the disease alkaptonuria, the results in a very dark urine and arthritis later in life. By studying family histories of the condition he was able to show that it was recessively inherited. He was then the first to propose the idea that some diseases were “inborn errors of metabolism” and that one gene produces one protein”. This is the molecular basis of inheritance
Later Garrod served in Malta as medical consultant to the army in the first world war. A war that would see him lose 2 of his 3 sons, with his 3rd son dying shortly later from the Spanish Flu.

It was moldy bread that finally proved Garrod’s hypothesis of one gene one protein being. In 1941 George Beadle and Edward Tatum saw that exposing molds to radiation reduced their growth as a result of inhibiting their ability to produce essential nutrients. However, providing the mutated mold with a specific supplement allowed to mold to continue growing again. From this is was concluded that each mutation must inactivate the enzyme (protein) needed to synthesize the nutrient. Thus, one gene carries the directions for making one protein.

‘For each chromosome contributed by the sperm there is a corresponding chromosome contributed by the egg, there are two chromosomes of each kind, which together constitute a pair.’ – Thomas Hunt Morgan
Thomas Hunt Morgan
Thomas Hunt Morgan showed that chromosomes carry genes so forming the basis of heredity.
Born in Kentucky in 1866 to a distinguished southern family whose was the great-grandfather was Francis Scott Key, the author of the “Star Spangled Banner
Morgan’s research involved the study of mutations in the fruit fly Drosophila. One day he came across one male fly in one of his culture bottles that had white-eyes. Simply out of curiosity he bred the fly with normal (red-eyed) females. This produced offspring that all had red-eyes. Crossing the offspring (brother–sister matings) produced an F2 generation with some white-eyed flies, all of which were males. From this he postulated that the X-chromosome carries a number of discrete hereditary units he described as genes (a term introduced by Wilhelm Johannsen in 1909). He found a number of other genetic variations that were also sex-linked and usually inherited together, leading him to further conclude that genes were arranged in a linear fashion on chromosomes.
In 1933 he won the Nobel prize for his work that was followed by further Nobel prizes for a number of his students including George Wells Beadle and Hermann Joseph Muller.

Frederick Griffith
Frederick Griffith was employed by the British government during World War II to develop a vaccine against “Spanish flu “. Working with the tiniest of budgets it was said that he “could do more with a kerosene tin and a primus stove than most men could do with a palace“.
His amazing discovery was that a harmless strain of Streptococcus pneumoniae could be made lethal again after being exposed to heat-killed virulent strains. This lethal virulent strain had a polysaccharide capsule that gave them a smooth surface (hence the name “S” strain) while a second rough “R” strain – did not cause pneumonia when injected into mice, since it lacked a capsule. Dead S strain bacteria, killed by heating, are not lethal to mice. But when this dead S strain is mixed with live R strain bacteria and injected the mouse dies. When he isolated the bacteria from this dead mouse he saw that all the live bacteria were of the S strain; the R bacteria had acquired capsules from the dead S bacteria. This led him to hypothesis that some “transforming principle” from the heat-killed S strain converted the R strain into the virulent S strain.
Killed during the London Blitz in 1941 along with director of the laboratory, William M. Scott, their deaths received hardly any mention.

‘C (somatic) carbohydrate, nucleoproteins, free nucleic acids of both the yeast [RNA] and thymus [DNA] types, lipids and other cell constituents. Try to find in that complex mixture the active principle..!! Try to isolate and chemically identify the par- ticular [transforming] substance….!Some job–full of heartaches and heartbreaks. But at last perhaps we have it….’ – Oswald Avery
Avery, McLoud & McCartey
Three scientists, Oswald Avery, Colin MacLeod, and Maclyn McCarty, managed to show that Frederick Griffith’s transforming factor was in fact DNA, i.e. DNA is the heritable substance.
At first, Avery refused to believe Griffith’s results that actually challenged his own research on pneumococcal capsules – how could a rough capsule be converted into a smooth one? However, he soon confirmed Griffith’s results and set about trying to purify this mysterious “transforming principle” – a substance that could cause a heritable change of bacterial cells.
They extracted from Streptococcus pneumoniae S (containing a capsule) bacteria purified DNA, proteins and other materials and mixed R bacteria (lacking a capsule) with these different materials, and only those mixed with DNA were transformed into S bacteria. Therefore, DNA is the “transforming factor” and not proteins or other materials.
Amazingly, not everyone was convinced by the experiments of Avery’s, MacLeod’s and McCarty’s as it was still widely assumed that genetic information was carried in protein. Firstly, due to Levene’s influential “tetranucleotide hypothesis”, many condsidered DNA to be a “stupid molecule,” made up of a repeat of the four chemical bases without any variations. Secondly, few biologists thought that genetics could be applied to bacteria, since they lacked chromosomes and sexual reproduction. Although the experimental findings of their experiment were quickly and independently a number of scientists still considered that protein contaminants were responsible the results. It was not be until the experiments of Hershey and Chase (1952) that DNA was finally proved and accepted to be the genetic material.
As Avery, MacLeod and McCarty concluded in their 1944 paper: “If the results of the present study on the chemical nature of the transforming principle are con- firmed, the nucleic acids must be regarded as possessing biological specificity the chemical basis of which is as yet undetermined.”
This blazing discovery, in many ways, radically changed the direction of genetics driving research to study DNA structure and sequences. Sadly the work was neglected by the Nobel Foundation, which did later expressed public regret.
It is a universally recognized omission of the Nobel committee since their work was a blazing discovery that changed everything. For more about this topic see in the link section “The Nobel Prize Omission

Phoebus Levene
Born in Sagor, Russia, Phoebus Levene emigrated to the U.S with his family to escape growing anti-Semitism in Russia. He split his time between medical practice and research on the chemical structure of sugars before leading a biochemical laboratory at the Rockefeller Institute. Here he identifies the components of DNA as a purine or pyrimidine nucleobase (simply called a base), a pentose sugar and a phosphate group base. These were then linked together in the order phosphate-sugar-base to form units and that DNA consisted of a string of these nucleotide units linked together. However, he though that a DNA molecule contained only four of these units linked together in a repeated manner, i.e. a tetranucleotide. Furthermore, he considered such a simple sequence could not allow DNA any role in coding for anything. This was later to be proved wrong by Chargaff.

‘Science is wonderfully equipped to answer the question “How?” but it gets terribly confused when you ask the question “Why?”‘- Erwin Chargaff
Erwin Chargaff
Erwin Chargaff proved that DNA was not organized into a regular 4 repeat pattern. Analysing the components of DNA he found a pattern where the number of guanines residues equalled the number of cytosines and the number of adenines was the same as the number of thymines. Furthermore, he saw that the relative amounts of the Guanines/Cytosines and Adenines/Thymines varied among different species though remained the same among individuals of the same species, i.e. DNA sequences are variable and species specific.
Chargaff actually mentioned some of his newest results to Crick and Watson a year before they published their model of DNA structure – much to his chagrin! In fact, after they received the Nobel Prize Chargaff withdrew from his lab and wrote to scientists all over the world about his exclusion, taking it a little less graciously than Rosalind Franklin or Oswald Avery!

‘While the biological properties of deoxypentose nucleic acid suggest a molecularstructure containing great complexity, X-ray diffraction studies described here… show the basic molecular configuration has great simplicity.’– Rosalind Franklin
Rosalind Franklin
Rosalind Franklin developed X-ray crystallography – a technique of determining a molecule’s three-dimensional structure by analyzing the X-ray diffraction patterns of crystals made up of the molecule in question – to study DNA. After 100 hours of X-ray exposure from a machine Dr. Franklin herself she and a student of hers, Raymond Gosling, were finally able to produce the famous “photograph 51” revealing the structure while at King’s College in London.
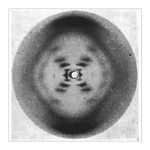
This allowed her to derive a number of important basic facts about DNA’s structure: the sugar-phosphate backbone of DNA lies on the outside of the molecule (not the inside as was previously thought); has a helical structure of two DNA strands (not three as proposed in competing theories); she was also able to provide quantitative details about the shape and size of the double helix.
The vital piece of the puzzle still eluded her was how the bases paired on the inside of the helix; this remained for Watson and Crick to discover.
After her boss at King’s College presented Franklin’s data and unpublished conclusions at a seminar a colleague of hers, Maurice Wilkins showed her photograph 51 to Watson and Crick without her knowledge. This helped to allow Watson and Crick to put all the pieces of the puzzle together, publishing their model for the structure of DNA appeared in the journal Nature in April, 1953, alongside Franklin’s own report.
Watson, Crick and Wilkins went on to win the Nobel Prize in 1962. By this time Rosalind had died of ovarian cancer 4 years earlier, at age 37. Whether this may have been a result of the radiation exposure from her work, or due to her own genetic makeup is unknown. Although, what is well remembered is that she behaved graciously and certainly showed no signs of bitterness.
Both Watson and Crick have since made it abundantly clear that they could not have discovered the structure of DNA without her work. Because the Nobel Prize is not awarded posthumously, Rosalind Franklin joins the number of unrewarded scientists in the DNA race.


‘It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material.’– Final remark by Watson and Crick in the Nature paper announcing discovery of the structure of DNA.
Watson & Crick
Francis Crick and James Watson knew DNA contained equal ratios of C and G residues and A and T residues. They also accepted from Rosalind Franklin’s X-ray data that DNA must consist of two anti-parallel chains and the backbone had to be on the outside. They considered that bases might be on the inside of the spiral. But it was hard to see how an irregular composition of bases could form a regular structure. They therefore ordered exact metal cut-outs from the bases to make a new model.
But this would take time! So Watson instead cutout cardboard templates of the four DNA bases. Playing with them on his cramped desk at Cambridge University, he initially tried to fit like with like i.e. C with C and A with A. but nothing worked. Suddenly, he noticed that an A-T pair lined up on his desk was identical in shape to a G-C pair. Immediately he saw how these could be the equal steps of DNA’s spiral staircase. Each of the four letters on one chain is matched by its opposite on the other; when the chains separate, each is the template for a new chain of exactly matching sequence.
Meselson & Stahl
Matthew Meselson and Franklin Stahl worked on an experiment to prove the semiconservative DNA replication hyposthesis proposed by James Watson and Francis Crick.
Often described the most beautiful experiment in biology they used isotopes of Nitrogen to differentially label the old and the newly replicated stands of DNA.

To do this they cultured E. coli for several generations in a medium with a heavy isotope of nitrogen, 15N. They then put the bacteria into a new medium with a lighter isotope of nitrogen, 14N. DNA samples were then taken from the bacteria; one after the first replication at 20 minutes and one after the second replication at 40 minutes. Using a centrifuge they were able to separate the DNA at different densities depending on the content of heavy 15N or the lighter 14N. The sample after 20 min revealed that all of the DNA had a consistent density as it was composed of one heavy strand and one light strand. The sample after 40 min contained some DNA at the same density as it was before (the hybrid DNA) and some DNA (the newly replicated DNA) that was less dense than before (14N). Thus the semiconservative model was proven to be correct!

‘It took less than an hour to make the atoms, a few hundred million years to make the stars and planets, but five billion years to make man! – George Gamow
George Gamow
George Gamow solved the riddle that the twenty combinations of four DNA bases, when taken three at a time, corresponds to the twenty amino acids used to form proteins, i.e. the idea of the triplet code.
Russian-born, he was a highly gifted theoretical physicist being elected a member of the Academy of Sciences of the USSR at age 28 — one of the youngest in the history – after a number of huge discoveries. However, increased oppression pushed him and his wife (who was also a physicist) to flee Russia. They firstly tried paddling a kayak for 36 hrs over the Black Sea to Turkey quickly followed by an attempt from Murmansk to Norway through the ice-cold Barents Sea – poor weather foiled both attempts. However, a year later, in 1933, they both managed to obtain permission to attend a physics Conference in Brussels, where they promptly defected and moved to the United States. There he lectured in physics and researched the Big Bang theory. It was only in his later years that Gamow started working in biology, applying his amazing reasoning and creativity to such startling effects.

The RNA Tie Club
Though the structure of DNA was now known, it was still not understood how everything fitted together. How did DNA code for proteins and how did RNA fit in?
The idea of James Watson and Russian physicist George Gamow, the RNA Tie Club was a scientific gentleman’s club of select members who could share their ideas and findings not yet ripe enough to be published in scientific journals.
Started in 1954 club met twice a year and consisted of 20 members representing each amino acid, with an additional four honorary members representing each nucleotide. Eight of these members went on to win Nobel Prizes. All members received woollen ties with an embroidered helix on them.
George Gamow, Physicist, ALA (Alanine)
Alexander Rich, Biochemist, ARG (arginine)
Paul Doty, Physical Chemist, ASP (Aspartic acid)
Robert Ledley, Mathematical Biophysicist, ASN (Asparagine)
Martynas Ycas, Biochemist, CYS (Cysteine)
Robley Williams, Electron Microscopist, GLU (Glutamic acid)
Alexander Dounce, Biochemist, GLN (Glutamine)
Richard Feynman, Theoretical Physicist, GLY (Glycine)
Melvin Calvin, Chemist, HIS (Histidine)
Norman Simons, Biochemist, ISO (Isoleucine)
Edward Telle r, Physicist, LEU (Leucine)
Erwin Chargaff, Biochemist, LYS (Lysine)
Nicholas Metropolis, Physicist, Mathematician, MET (Methionine)
Gunther Stent, Physical Chemist, PHE (Phenylalanine)
James Watson, Biologist, PRO (Proline)
Harold Gordon, Biologist, SER (Serine)
Leslie Orgel, Theoretical Chemist, THR (Threonine)
Max Delbrück, Theoretical Physicist, TRY (Tryptophan)
Francis Crick, Biologist, TYR (Tyrosine)
Sydney Brenner, Biologist, VAL (Valine)
Meetings generally involved cigars and alcohol allowing for bonding and close friendships. And indeed a number of influential ideas and concepts did arise. Gamow postulated that a nucleotide code consisting of three letters would be enough to define all 20 amino acids., i.e. the concept of the “codon”. Sydney Brenner provided experimental evidence for the codon while Francis Crick proposed the “Adaptor hypothesis” involving a molecule ferrying the amino acids around, and put them in the correct order corresponding to the nucleic acid sequence, these moleculaes were later found by Robert Holley and named transfer RNAs.
‘It’s always very refreshing to be able to just make a clean break, start over again with something you’re completely ignorant about.’– Seymour Benzer
Seymour Benzer
Like a number of the major players in the story of DNA, Seymour Benzer, was primarily a physicist. Beginning research on radar systems for the US government. Turning his interest to biology he studied viruses that infected E.coli bacteria. It was through his work that he found it was possible to delete a single base of the virus sequence with a special chemical. This would not be able produce a particular functional protein because the insertion or deletion of a single nucleotide caused a frameshift mutation. Mutants with two or four nucleotides inserted or deleted were also nonfunctional. However, the mutant strains could be made functional again by deletibg a total of three nucleotides. This proved that the genetic code uses a codon of three DNA bases that corresponds to an amino acid.
Following his succeses with viruses Benzer switched to the fruit fly Drosophila studying the genetic basis of animal behavior and again pioneering a field, this time working with mutants of Drosophila to study neurogenetics.

Marshall Nirenberg
Marshall Nirenberg, not a member of the RNA tie club, nevertheless had interest to try to crack the DNA/RNA code. To do this he hit upon a brilliant idea. He produced RNA consisting only of one base – uracil (a nucleotide specific only to RNA). Thy added this to a mixture containing the cellular machinery for protein synthesis, extracted from E.coli (i.e. they destroyed the cell membranes of E.coli and removed the RNA and DNA). They then added the 20 amino acids, but being sure to radiolabel only one of them. They consequently found that in the extract containing the radioactively labeled phenylalanine, the resulting protein was also radioactive leading them to conclude that that they had found the genetic code for phenylalanine: UUU (three uracil bases in a row) on RNA.
Within a few years, his research team had performed similar experiments and found that three-base repeats of adenosine (AAA) produced the amino acid lysine, and cytosine repeats (CCC) produced proline.
This finding led to the race to discover the whole genetic code for all 50 codes and their respective 20 amino acids. He was awarded the Nobel prize in 1968 for this feat.
The Coding Race
Faced with the possibility of helping the first NIH scientist win a Nobel prize, many NIH scientists put aside their own work to help Nirenberg in deciphering the mRNA codons for amino acids. Dr. DeWitt Stetten, Jr., director of the National Institute of Arthritis and Metabolic Diseases, called this period of collaboration “NIH’s finest hour.
